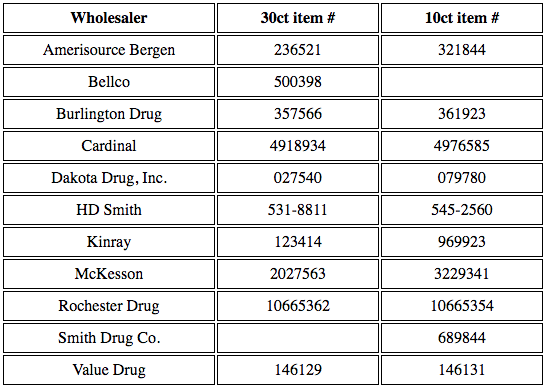
Hospitals & Pharmacists

Dear Hospital Pharmacist or NICU Provider:
FloraTummys Probiotic Sprinkles for Kids has been safely used in preterm infants in the NICU for over 7 years. Most protocols include mixing one FloraTummys packet with 3ml of breastmilk or formula during feeding or NG Tube with normal saline. For list of hospitals and protocols contact frank@floratummys.com or call 201-819-7643.
UPC Code: 8 52061 00400 4
NDC: 520610004-00
- Individual bar-coded powder packets
- 5 billion CFU: B. lactis & L. acidophilus
- Prebiotic (FOS)
- Non-Dairy, Gluten & Sugar-Free
- Free of 8 Top Food allergens
- Completely dissolves in liquids
- Won’t clog NG Tubes (see mix test vs Florastor & Culturelle)
- Made in the USA in a NSF GMP Certified Facility
“…our NEC rate dropped from 14.6% to 3.4% during the six months of probiotic use.”
From a Clinical NICU Pharm, D. using FloraTummys Feb 28, 2017
New Floratummys Probiotic Quick Dissolve Tablets: To Help Reduce Cross Contamination of Probiotics

UPC Code: 8 52061 00410 3
NDC #: 520610004-10
- Quickly & Completely Dissolves
- Can Help Reduce Cross Contamination
- Won’t Clog NG Tubes
- Individual Bar-Coded Packets
- Contains both B. infantis & L. reuteri & FOS
- Made in the USA in a NSF GMP Certified Facility
-
†† B. infantis is a protective bacteria historically found in infant gut microbiome which consumes HMO’s and produces lactate and acetate, which can protect against the growth of enteropathogens.
“We tested it and it took about 1 minute with some shaking to dissolve the tablet. We tried to dissolve the whole tablets in 1ml and 3ml of milks. The final products look good and easy to prepare! We couldn’t tell the difference if it was just EBM or EBM with probiotics. It didn’t give the grainy/viscous texture.”
Clinical NICU Pharm.D. after testing New FloraTummys® Quick Dissolve Tablets.
*Cross Contamination of Probiotics in Hospitals
The use of bulk probiotic powders and breaking open capsules can increase the likelihood of cross contamination in your hospital.
†NICU Studies have shown that control group infants who were not treated with probiotics were also colonized with probiotics . It can be deduced that Cross-Contamination or Cross Colonization does exist with probiotics.
FloraTummys Quick Dissolve Tablets in individual packets can help reduce cross contamination of probiotics in your hospital by reducing the chance of powder escaping into the air. The tablets are coated with a finish to protect the tablet from cracking and leaving residue.
Directions:
Shake or stir one tablet vigorously uing a closed container or syringe in at least 3ml of breastmilk, formula, or water for about two minutes or until completely dissolved before feeding.
†Gengaimuthu, K: The Cross Contamination (Cross Colonization) Phenomenon of Probiotic Use in NICUs: Neonatal and Pediatrics, International Modern Hospital, Dubai, ARE. May 2018. DOI: 10.7759/cureus.2691
Garland et aI.: The ProPrems Trial: investigating the effects of probiotics on late onset sepsis in very preterm infants. BMC Infectious Diseases 2011 11:210.
Kitajima H1 RTC: Early administration of Bifidobacterium breve to preterm infants: Arch Dis Child Fetal Neonatal Ed. 1997 Mar;76(2):F101-7.
Costeloe et aI. abstract at Neonatal Society 2004 Spring Meeting in the UK
NEC Society Symposium, April 2019, Ann Arbor, MI
Fukuda S et al.Nature. 2011,469 (7331) 543-547
The Cochrane Review: (24 NEC-Probiotic studies) The authors concluded:
“Enteral supplementation of probiotics prevents severe NEC and all cause mortality in preterm infants. Our updated review of available evidence strongly supports a change in practice. Head to head comparative studies are required to assess the most effective preparations, timing, and length of therapy to be utilized.”
“The administration of a mixture of probiotics (nine trials) significantly reduced the incidence of mortality (RR 0.62, 95% CI 0.47 to 0.81). The administration of Lactobacillus species alone (four trials), bifidobactirium species alone (two trials), or Saccharomycesboulardii alone (one trial) did not reduce mortality…”
2016 Pediatric Pharmacy Advocacy Group (PPAG) Probiotics-NEC Review. Recommendations were to use:
- Multi-strain probiotics with both lactobacillus and bifidobacterium
- At least 1 billion CFU
- Powder packets vs bulk powder to reduce risk of cross contamination
Results: “…The use of dual-strain probiotics significantly reduced the risk of NEC (HR = 0.48; 95% CI = 0.38-0.62), overall mortality (HR = 0.60, 95% CI = 0.44-0.83), mortality after NEC (HR = 0.51, 95% CI = 0.26-0.999) and nosocomial BSI (HR = 0.89, 95% CI = 0.81-0.98).
These effects were even more pronounced in the subgroup analysis of preterm infants with birth weights below 1,000 g.”
Denkel LA1, Schwab F1, Garten L2, Geffers C1, Gastmeier P1, Piening B1.Institute of Hygiene and Environmental Medicine, Charité Universitätsmedizin Berlin, Berlin, Germany.2 Dept. of Neonatology, Charité Universitätsmedizin Berlin, Berlin, Germany.
Probiotics and Necrotizing Enterocolitis: Finding the Missing Pieces of the Probiotic Puzzle
“NEC rate should be one of the first determinants when considering the use of probiotics for the prevention of NEC in the NICU.”
Sherry A. Luedtke, PharmD, Jacob T. Yang, PharmD, and Heather E. Wild, PharmD
Texas Tech University Health Sciences Center School of Pharmacy, Amarillo, Texas